|
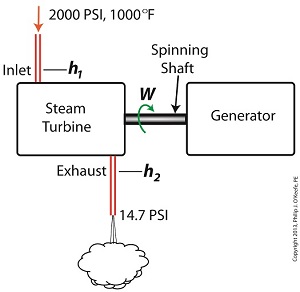
Last time we learned that the amount of useful work, W, that a steam turbine performs is calculated by taking the difference between the enthalpy of the steam entering and then leaving the turbine. And in an earlier blog we learned that a vacuum is created in the condenser when condensate is formed. This vacuum acts to lower the pressure of turbine exhaust, and in so doing also lowers the enthalpy of the exhaust steam. Putting these facts together we are able to generate data which demonstrates how the condenser increases the amount of work produced by the turbine. To better gauge the effects of a condenser, let’s look at the differences between its being present and not present. Let’s first take a look at how much work is produced by a steam turbine without a condenser. The steam entering the turbine inlet has a pressure of 2000 pounds per square inch (PSI) and a temperature of 1000°F. Knowing these turbine inlet conditions, we can go to the steam tables in any thermodynamics book to find the enthalpy, h1. Titles such as Fundamentals of Classical Thermodynamics by Gordon J. Van Wylen and Richard E. Sonntag list enthalpy values over a wide range of temperatures and pressures. For our example this volume tells us that, h1 = 1474 BTU/lb where BTU stands for British Thermal Units, a unit of measurement used to quantify the energy contained within steam or water, in our case the water to steam cycle inside a power plant. Technically speaking, a BTU is the amount of heat energy required to raise the temperature of one pound of water by one degree Fahrenheit. The term lb should be a familiar one, it’s the standard abbreviation used for pound, so enthalpy is the measurement of the amount of energy per pound of steam flowing through, in this case, the turbine. Since there is no condenser attached to the steam turbine’s exhaust in our illustration, the turbine discharges its spent steam into the surrounding atmosphere. The atmosphere in our scenario exists at 14.7 PSI because our power plant happens to be at sea level. Knowing these facts, the steam tables inform us that the value of the exhausted steam’s enthalpy, h2, is: h2 = 1015 BTU/lb Combining the two equations we are able to calculate the useful work the turbine is able to perform as: W = h1 – h2 = 1474 BTU/lb – 1015 BTU/lb = 459 BTU/lb This equation tells us that for every pound of steam flowing through it, the turbine converts 459 BTUs of the steam’s heat energy into mechanical energy to run the electrical generator. Next week we’ll connect a condenser to the steam turbine to see how its efficiency can be improved.
________________________________________ |
Posts Tagged ‘water’
Enthalpy Values in the Absence of a Condenser
Tuesday, November 26th, 2013Condensation Inside the Steam Turbine
Sunday, September 8th, 2013|
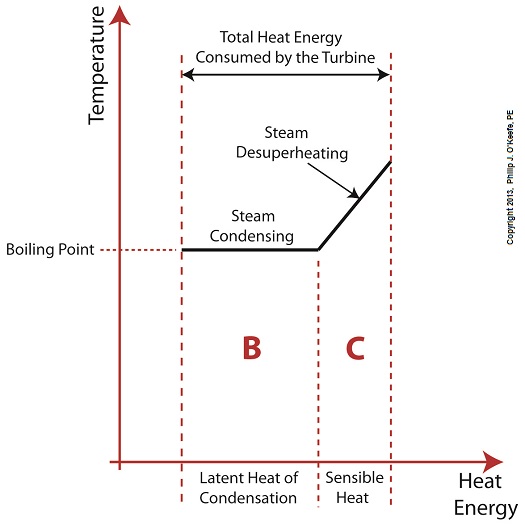
Did you know that water droplets traveling at high velocity can take on the force of bullets? It can happen, particularly within steam turbines at a power plant during the process of condensation, where steam transforms back into water. The last couple of weeks in this blog series we’ve been talking about the steam and water cycle within electric utility power plants, how heat energy is added to water during the boiling process, and how turbines run on the sensible heat energy that lies within the superheated steam vapor supplied by boilers and superheaters. We learned that without a superheater there is a very real possibility that the steam’s temperature can fall to mere boiling point. When steam returns to boiling point temperature an undesirable situation is created. The steam begins to condense into water within the turbine. To understand how this happens, let’s return to our graph from last week. It illustrates the situation when there’s no superheater present in the power plant’s steam cycle. Figure 1
After consuming all the sensible heat energy in phase C in Figure 1, the only heat energy which remains available to the turbine is the latent heat energy in phase B. If you recall from past blog articles, latent heat energy is the energy added to the boiler water to initiate the building of steam. As the turbine consumes this final source of heat energy, the steam begins a process of condensation while it flows through the turbine. You can think of condensing as a process that is opposite to boiling. During condensation, steam changes back into water as latent heat energy is consumed by the turbine. When the condensing process is in progress, the temperature in phase B remains at boiling point, but instead of pure steam flowing through the turbine, the steam will now include water droplets, a dangerous mixture. As steam flows through the progressive chambers of turbine blades, more of its latent heat energy is consumed and increasingly more steam turns back into water as the number of water droplets increases. Figure 2 – Water Droplets Forming in the Turbine
The danger comes in when you consider that the steam/water droplet mixture is flying through the turbine at hundreds of miles per hour. At these high speeds water droplets take on the force of machine gun bullets. That’s because they act more like a solid than a liquid due to their incompressible state. In other words, under great pressure and at high speed water droplets don’t just harmlessly splash around. They hit hard and cause damage to rapidly spinning turbine blades. Without a working turbine, the generator will grind to a halt. So how do we supply the energy hungry turbine with the energy contained within high temperature superheated steam in sufficient quantities to keep things going? We’ll talk more about the superheater, its function and construction, next week.
________________________________________ |
Heat Energy Within the Power Plant— Water and Steam Cycle, Part 2
Wednesday, August 14th, 2013|
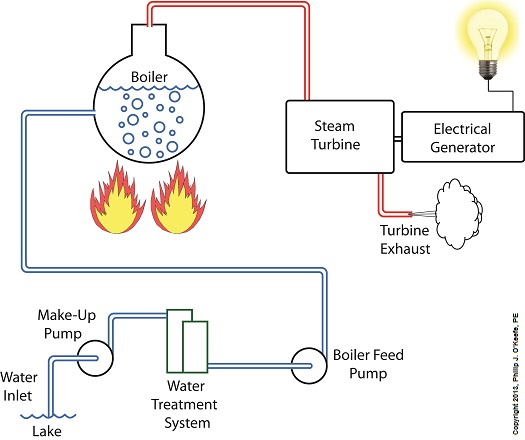
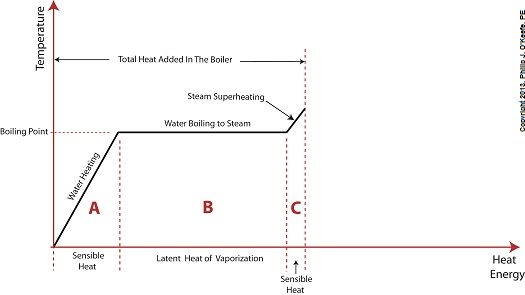
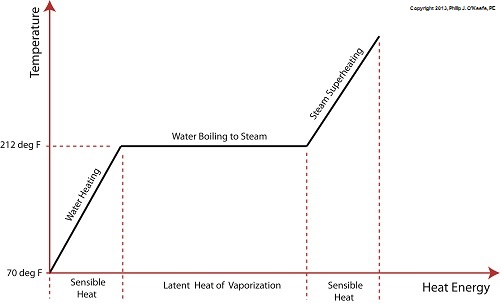
Last time we learned that electric utility power plants must have water treatment systems in place to remove contaminants from incoming feed water before it can be used. This clarified water is then fed to a boiler by the boiler feed pump as shown below. As it stands this setup will work to provide electricity, however in this state it’s both inefficient and wasteful. We’ll see why in a minute. Boilers, as their name implies, do a great job of heating water to boiling point to produce steam. They do this by adding the heat energy produced by burning fuel, such as coal, to water, then steam. We learned in earlier blogs in this series that the energy used to heat water to boiling point temperature is known as sensible heat, whereas the heat energy used to produce steam is known as latent heat. The key distinction between these two phases is that during sensible heating there is a rise in temperature, during latent heating there is not. For a review on this, see this blog article. When water starts to heat inside the boiler, sensible heat energy is said to be added. This is represented by phase A of the graph below. During A, heat energy will raise the temperature of the water to boiling point. As the water continues to boil in phase B, water is transforming into steam. During this phase latent heat energy is said to be added, and the temperature will remain at boiling point. In phase C something new takes place. The temperature rises beyond boiling point and only steam is present. This is known as superheated steam. For example, if the boiler pressure is at 1,500 pounds per square inch, steam becomes superheated at temperatures greater than 600°F. Unfortunately, boilers alone do a poor job of superheating steam, that is, continuing to raise the temperature of the steam present in phase C. This is evident by the fact that phase C is quite small in comparison to phases A and B before it. This inefficiency in producing ample amounts of superheated steam results in a small amount of useful energy being provided to the turbine down the line, which is bad, because steam turbines require exclusively superheated steam to run the generator. Next time we’ll see how to provide our steam turbine with more of what it needs to run the generator, more superheated steam. ___________________________________________
|
Heat Energy Within the Power Plant—The Power Behind the Turbines
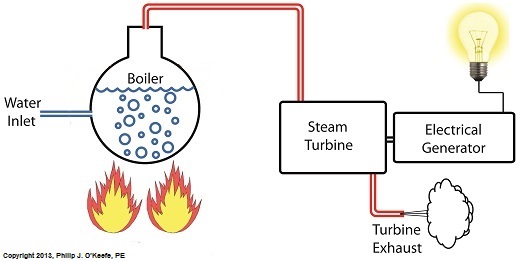
Monday, July 29th, 2013|
Last time we discovered that the boiling point of water varies. It’s dependent upon the amount of pressure exerted on its surface, which varies due to a variety of reasons, including where it is in relation to sea level. Before we see what happens under higher than atmospheric pressures, such as exist in an electric utility power plant boiler, let’s cover some basics. In the power plant, water is heated in a boiler specifically to produce steam, unlike our tea kettle where the primary purpose is to produce hot water. The steam produced is used to spin turbine generators, which in turn generate electricity, as I explained in a previous blog on steam turbines. Unlike a tea kettle, which is open to the atmosphere on your kitchen stove, the boiler in a power plant is an enclosed, reinforced steel vessel. See illustration below. The reinforced steel boiler vessel is designed to withstand great internal pressure as temperatures rise within. In addition to providing a safety feature, the enclosed space provides a sheltered environment for collecting steam so it can later be put to use spinning power generating turbines down the line. In other words, surface water inside the boiler is closed off from the surrounding atmosphere, allowing its internal pressure to build for our specific purposes. As heat energy is added to water within the boiler, the water boils and steam bubbles break out from its surface, filling the empty space above the surface with pressurized steam. This steam will try to expand here, but it can’t, because it’s being constrained by the reinforced steel vessel within which it is enclosed. Instead, steam pressure builds up on the surface of the water inside the boiler until it is high enough to be released through an attached pipe which is connected to a nearby turbine. We’ll talk more about this pent-up energy and how it is put to use within the power plant in next week’s blog. ___________________________________________
|
Forms of Heat Energy – Boiling Water and Atmospheric Pressure
Sunday, July 21st, 2013|
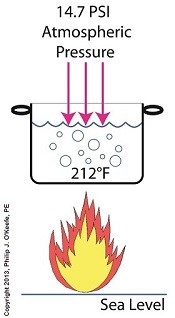
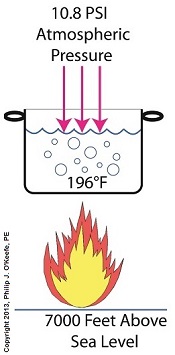
If you’ve ever baked from a pre-packaged cake or cookie mix, you’ve probably noticed the warning that baking times will vary. That’s because the elevation of the area in which you’re doing the baking makes a difference in the baking time required. Living in New Orleans? Then you’re at or below sea level. In Colorado? Then you’re above sea level. Your cake will be in the oven more or less time at the prescribed temp, depending on your location. Last time we learned how the heat energy absorbed by water determines whether it exists in one of the three states of matter, gas, liquid, or solid. We also learned that at the atmospheric pressure present at sea level, which is about 14.7 pounds per square inch (PSI), the boiling point of water is 212°F. At sea level there are 14.7 pounds of air pressure bearing down on every square inch of water surface. Again, I said sea level for a reason. The boiling point of water, just like cake batter baking times, is dependent upon the amount of pressure that’s being exerted on its surface from the surrounding atmosphere. When heat energy is absorbed, it causes the water or cake batter molecules to move around. In fact, the temperature measured is a reflection of this molecular movement. As more heat energy is absorbed, the molecules move more and more rapidly, causing temperature to increase. When the water temperature in our tea kettle reaches its boiling point of 212°F at sea level, the steam molecules in the bubbles that form have enough energy to overcome the atmospheric pressure on the surface of the water. They become airborne and escape in the form of steam. If we’re up in the Rockies at say an altitude of 7000 feet above sea level, the atmospheric pressure is only about 10.8 PSI. There’s just less air up there. That means there’s less air pressure resting upon the surface of the water, so it’s far easier for steam molecules to form into bubbles and leave the surface. As a result the boiling point is much lower in the Rockies than it is at sea level, 196°F versus 212°F. So what if the water was boiling in an environment that had even higher pressures exerted upon it than just atmospheric? We’ll see how to put this pent-up energy to good use next week, when we begin our discussion on how steam is used within electric utility power plants.
___________________________________________
|
Forms of Heat Energy – Sensible
Sunday, July 7th, 2013| In our house the whistle of a tea kettle is heard throughout the day, no matter the temp outside. So what produces that familiar high pitched sound?
When a tea kettle filled with room temperature water, say about 70°F, is heating on the stove top, the heat energy from the burner flame will transfer to the water in the kettle and its temperature will steadily rise. This heat energy that is absorbed by the water before it begins to boil is known as sensible heat in thermodynamics. To read more about thermodynamics, click on this hyperlink to one of my previous blog articles on the topic. So, why is it called sensible heat? It’s so named because it seems to make sense. The term was first used in the early 19th Century by some of the first engineers who were working on the development of boilers and steam engines to power factories and railways. Simply stated, it’s sensible to assume that the more heat you add to the water in the kettle, the more its temperature will rise. So how high will the temperature rise? Is there a point when it will cease to rise? Good questions. We’ll answer them next week, along with a discussion on another form of heat energy known as the latent heat of vaporization. ___________________________________________
|
Ground Fault Circuit Interrupters
Sunday, June 26th, 2011| I’ve been talking about how I was asked to be a subject matter expert for an upcoming series on The Discovery Channel titled Curious and Unusual Deaths. Most of the accidents discussed involved electrocutions, and in each case the electrocution occurred because the victim’s body, usually their hand, inadvertently contacted a source of current. When that happened their bodies essentially became like a wire, providing an unintended path for current to travel on its way to the ground. Why does it travel to the ground, you ask? Because electric current, by its very nature, always wants to flow along a conductor of electricity from a higher voltage to a lower voltage. The ground is the lowest voltage area on our planet. When electricity flows to ground along an unintended path it’s referred to as a “ground fault,” because that’s where the electricity is headed, to the ground, or Earth. By “fault” I mean that something in an electrical circuit is broken or not right, allowing the electrical current to leak out of the circuit along an unintended path, like through a person’s body.
For example, in one of the Curious and Unusual Deaths segments I was asked to explain how a fault in wiring caused electrical current to flow through a woman’s body to the ground that she was standing on. This happened when she unintentionally came in contact with a metal door that was, unbeknownst to her, electrically charged from an unanticipated source. The current was strong enough to cause her death. Where did the electric current originate from? Watch the program to find out, but I’m sure you’d never guess. To say that it was an unlikely source is an understatement. When ground faults pass through a person’s body, bad things often happen, ranging from a stinging shock to stopping your heart muscle to burning you from the inside out. The severity depends on a number of factors, including the strength of the current to the amount of time your body is exposed to it. It might surprise you to know that if your skin is wet at the time of contacting a current, you risk a greater chance of injury. Water, from most sources, contains dissolved minerals, making it a great conductor of electricity. But what exactly is electrical current? Scientifically speaking it’s the rate of flow of electrons through a conductor of electricity. Let’s take a closer look at a subject close to home, a power cord leading from a wall’s outlet to the electric motor in your kitchen hand mixer. That power cord contains two wires. In the electrical world one wire is said to be “hot” while the other is “neutral.” The mixer whirrs away while you whip up a batch of chocolate frosting because electrons flow into its motor from the outlet through the hot wire, causing the beaters to spin. The electrons then safely flow back out of the motor to the wall outlet through the neutral wire. Now normally the number of electrons flowing into the motor through the hot wire will basically equal the number flowing out through the neutral wire, and this is a good thing. When current flow going in equals current flow going out, we end up enjoying a delicious chocolate cake. Since the human body can conduct electricity, serious consequences may result if there is an electrical defect in our hand mixer that creates a ground fault through the operator’s body while they are using it. In that situation the flow of electrons coming into the mixer from the hot wire will begin to flow through the operator’s body rather than flowing through the neutral wire. The result is that the number of electrons flowing through the hot wire does not equal the flow of electrons flowing through the neutral wire. Electrons are leaking out of what should be a closed system, entering the operator’s body instead while on its way to find the ground. Next time we’ll look at a handy device called a Ground Fault Circuit Interrupter (GFCI) and how it keeps an eye on the flow of electrons, which in turn keeps us safe from being electrocuted. _____________________________________________ |